THE PURPLE STANDARD™
The Infrastructure Behind Every Purple Vial
Most companies sell products. We built a system.
The Purple Standard™ is our internal quality framework — a multi-layered sourcing, verification, and documentation protocol designed to protect molecular precision at every step.
This isn’t marketing language. It’s operational infrastructure.
When you see Purple Verified™, you’re seeing The Purple Standard™ at work.
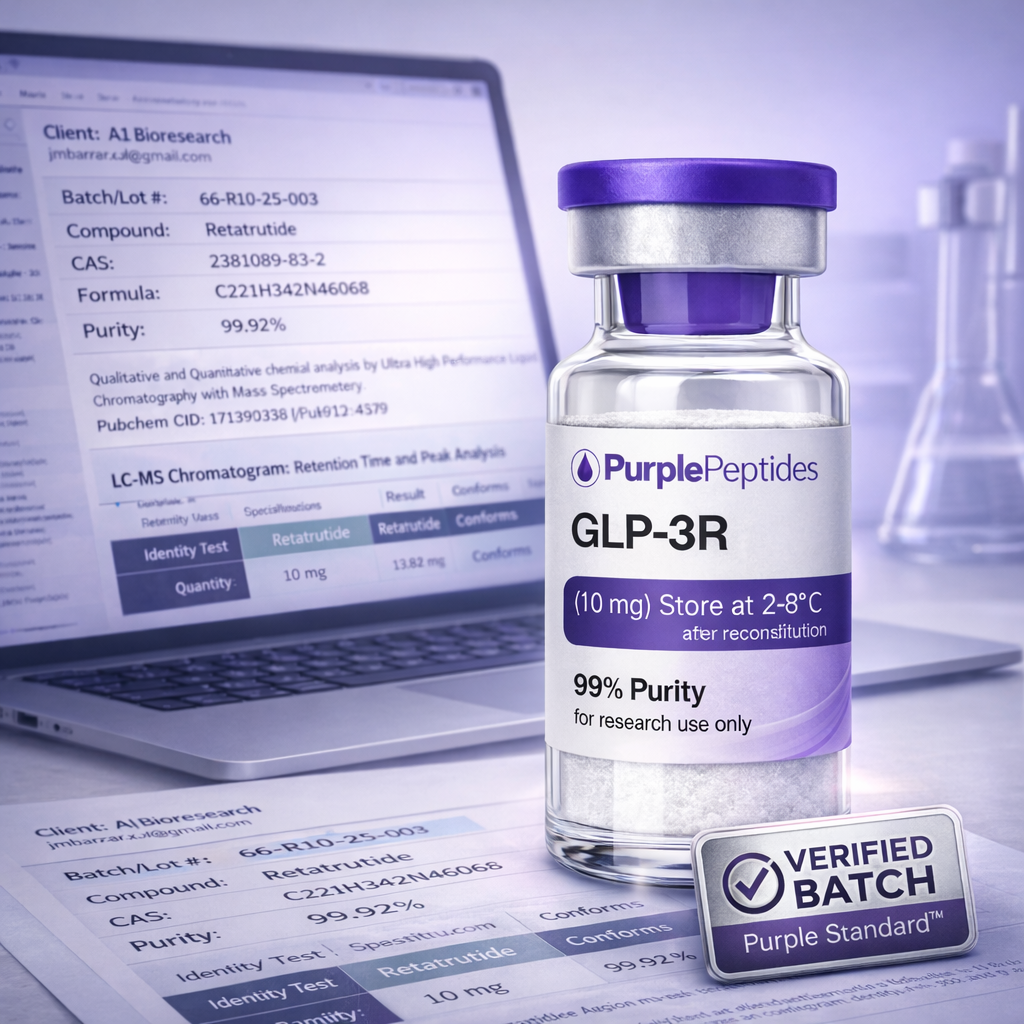
Standards built for longevity.
The peptide market moved fast. Standards didn’t. The Purple Standard™ exists to replace blind trust with documented proof — so every release is backed by infrastructure, not opinions.
How a batch becomes Purple Verified™
A release is earned — through identity confirmation, purity validation, documentation checks, and alignment across layers.
The system behind every release.
This framework exists to protect molecular integrity — and to ensure every Purple Verified™ batch is supported by documentation, not hype.
≥ 99% via HPLC + MS verification + sequence confirmation.
Manufacturer verification + independent third-party confirmation.
No mismatches, no gaps, no contaminant exceptions.


Not all “99%” claims are equal.
Many suppliers advertise high purity numbers without consistent validation or independent confirmation. Under The Purple Standard™, every peptide must meet:
One test is not enough.
Every batch undergoes two layers of verification:
- Identity confirmation
- Initial HPLC purity report
- Batch traceability documentation
- Confirmatory HPLC analysis
- Mass spectrometry verification
- Randomized cross-batch validation
- Impurity + contaminant screening


This is where most companies go quiet. We don’t.
A batch is rejected if:
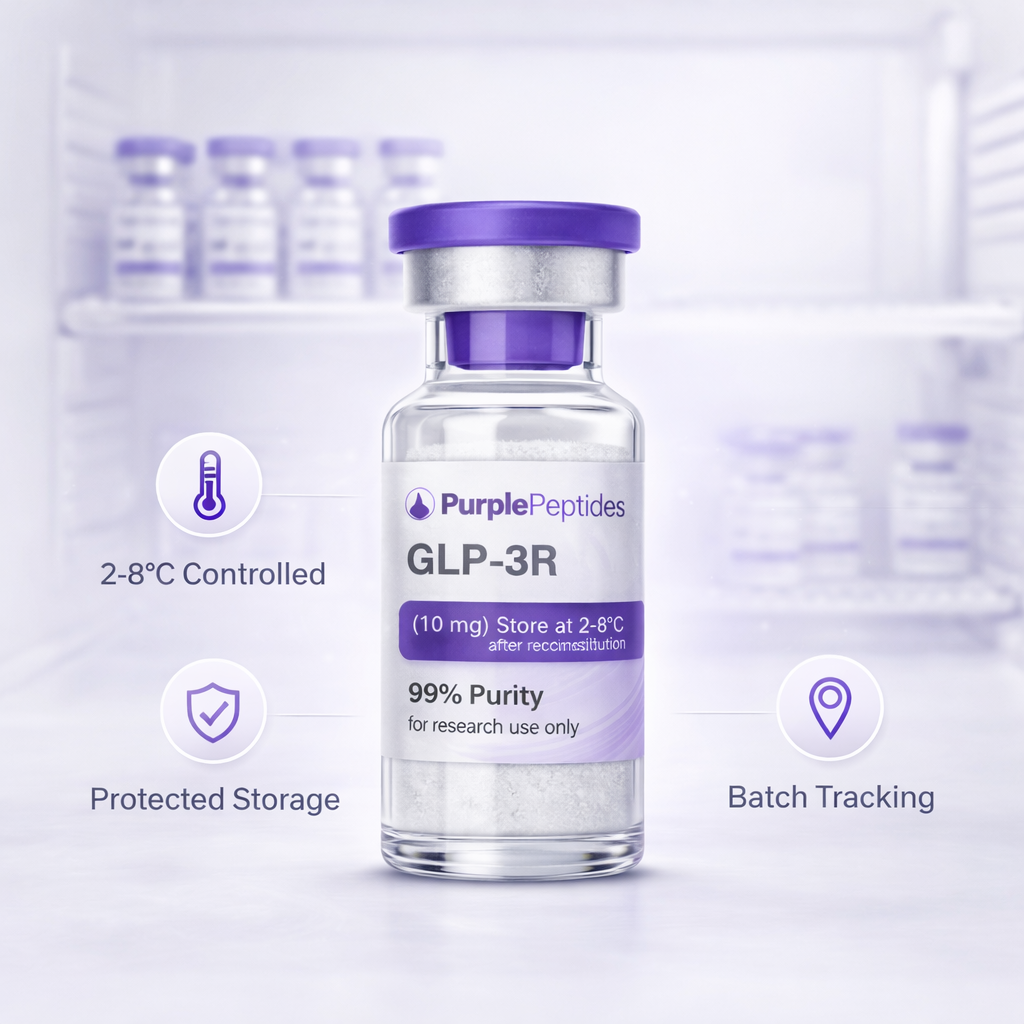
Molecular stability matters.
Peptides are sensitive compounds. Stability is protected through disciplined storage, controlled handling, and batch-level tracking.
Peptides require:
Our internal controls include:

Transparency builds trust.
Every product aligned with The Purple Standard™ includes:
Measured. Controlled. Documented. Protected.
This is the foundation underneath every Purple Verified™ release.
Not a Shopify hustle. Infrastructure.
When someone sees The Purple Standard™, they should think:
Brands that cut corners disappear. Brands that build standards endure. The Purple Standard™ is not a campaign. It is our operating philosophy — and the infrastructure behind everything we release.
- Cut corners to increase margins
- Skip independent verification
- Hide sourcing information
- Make reckless claims
- Prioritize purity
- Demand documentation
- Protect the integrity of our brand
- Respect the intelligence of our customers
